SPARC
Equity Metrics
May 8, 2026
Sun Pharma Advanced Research Company Limited
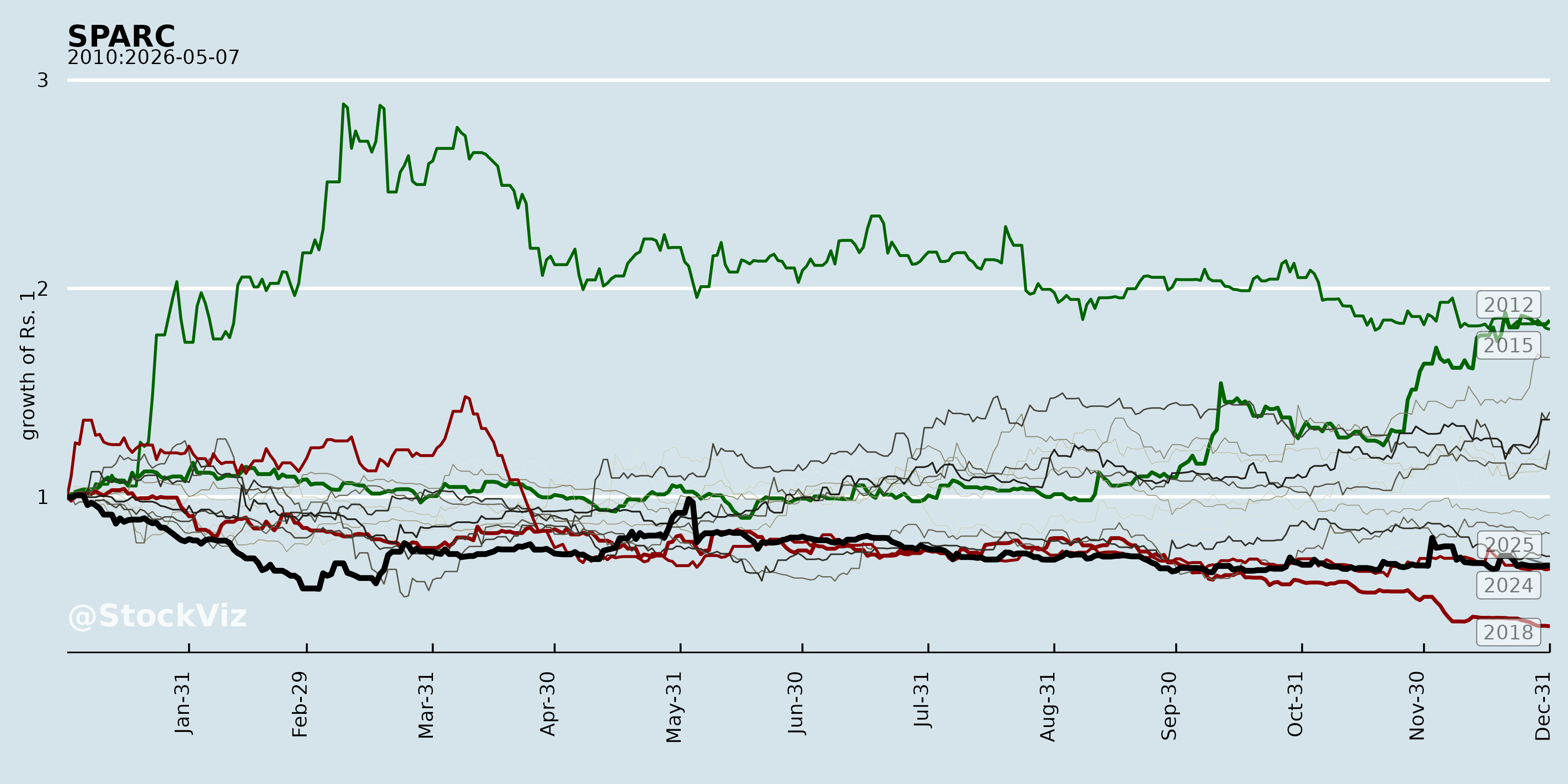
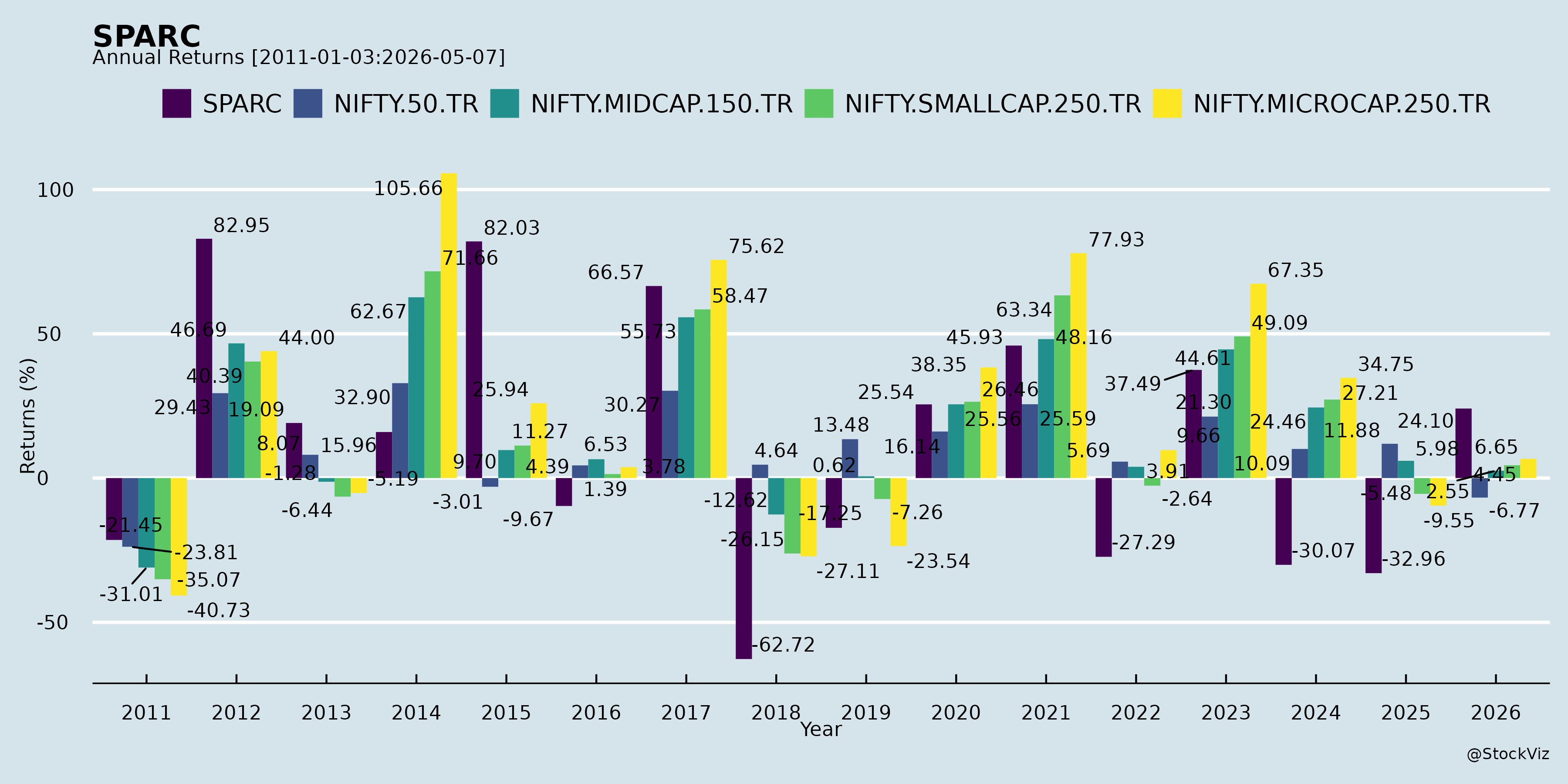
Annual Returns
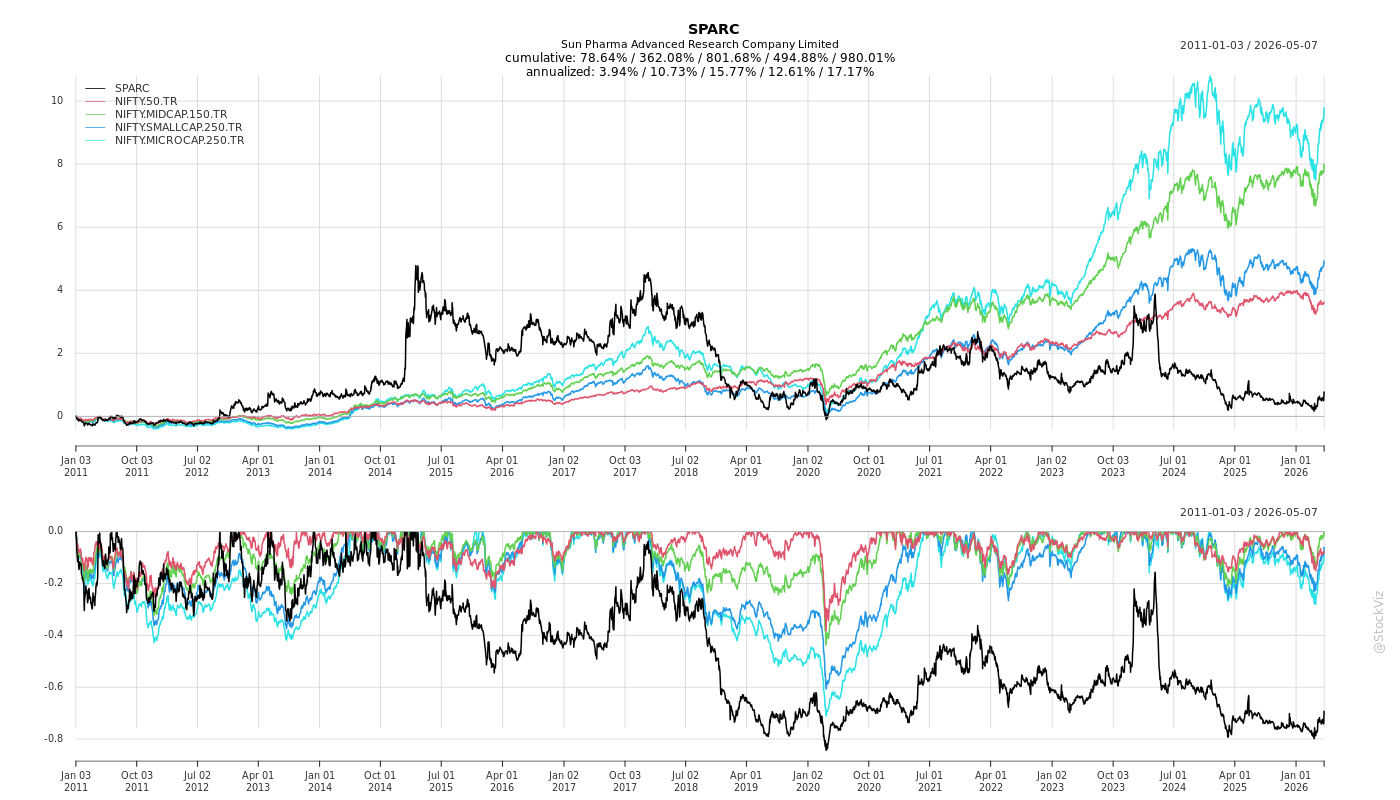
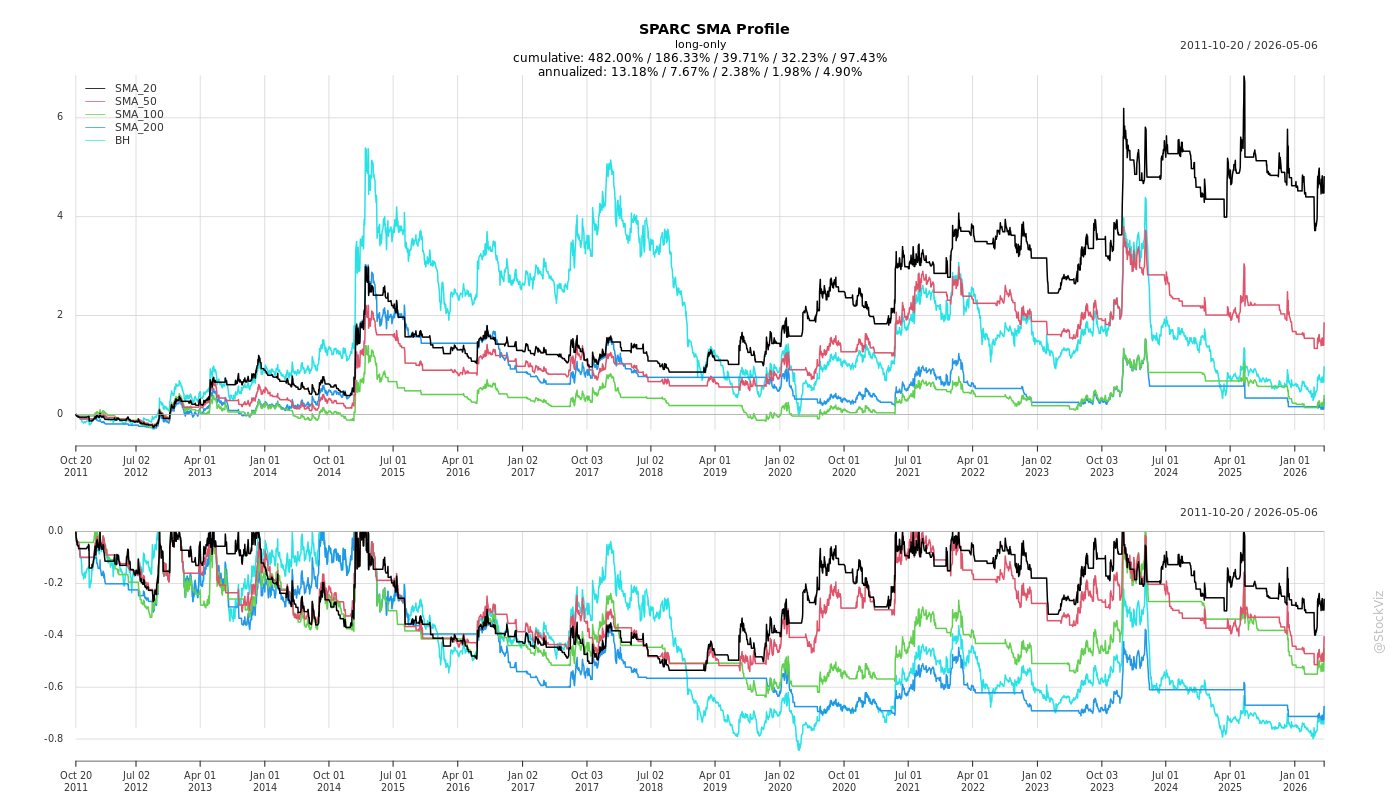
Cumulative Returns and Drawdowns
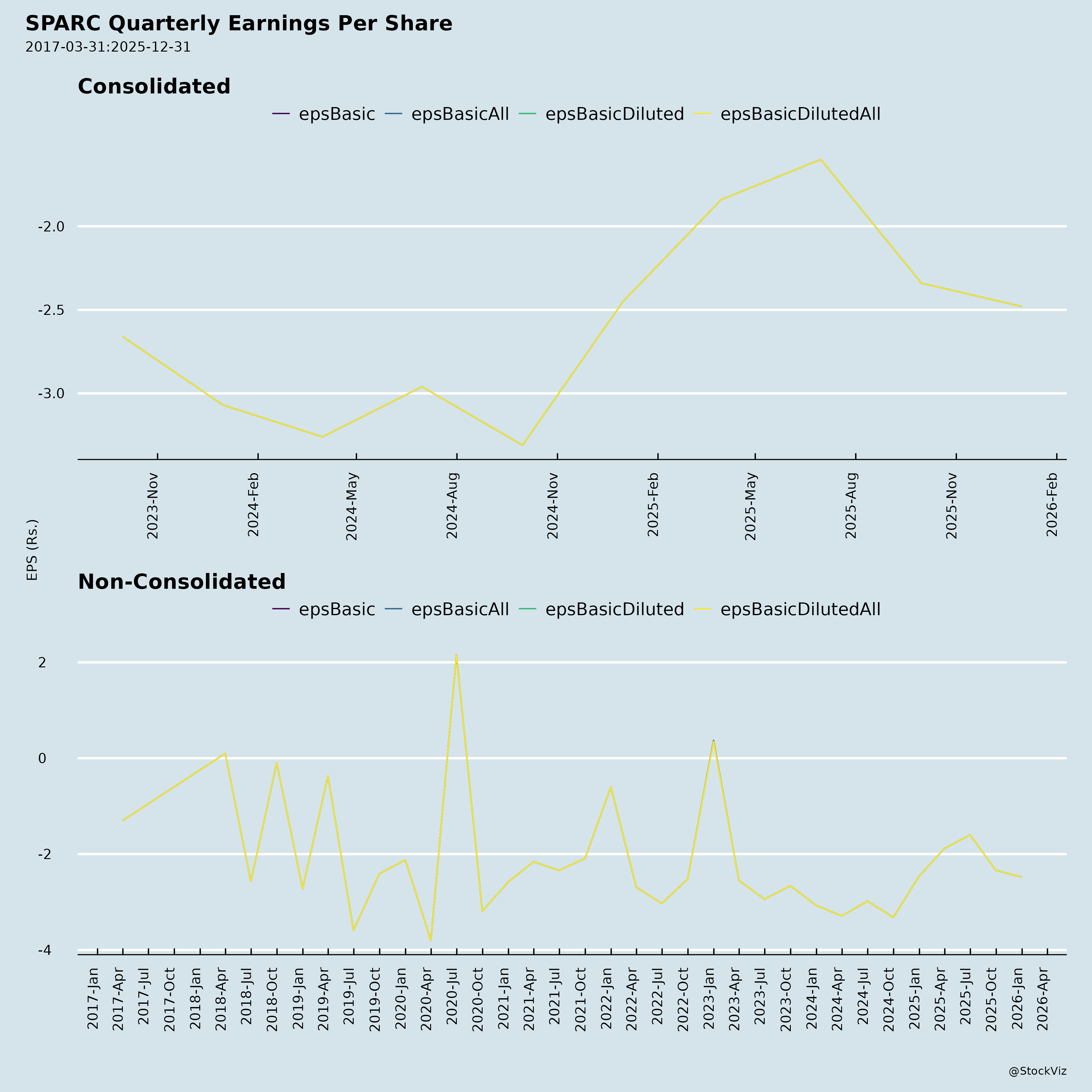
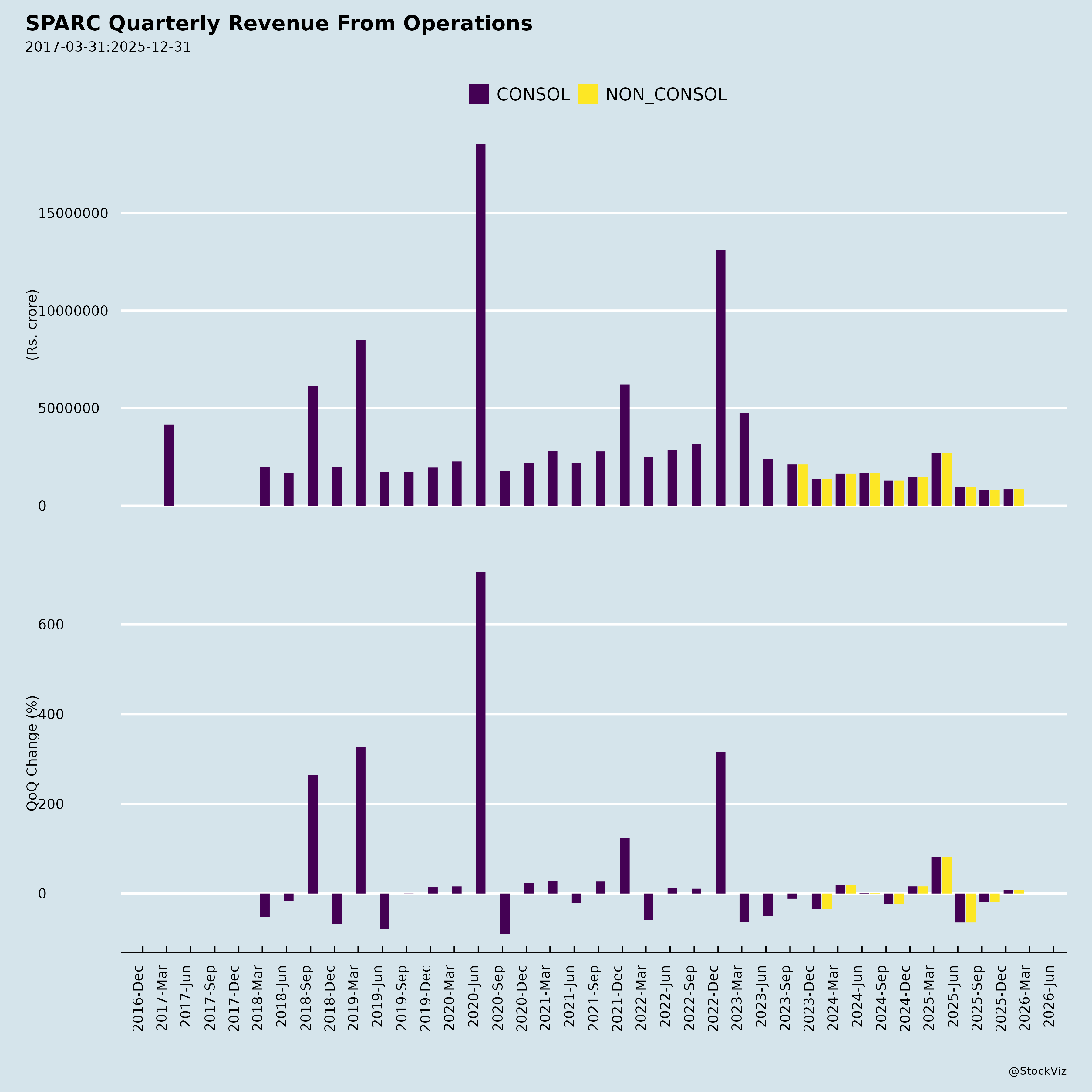
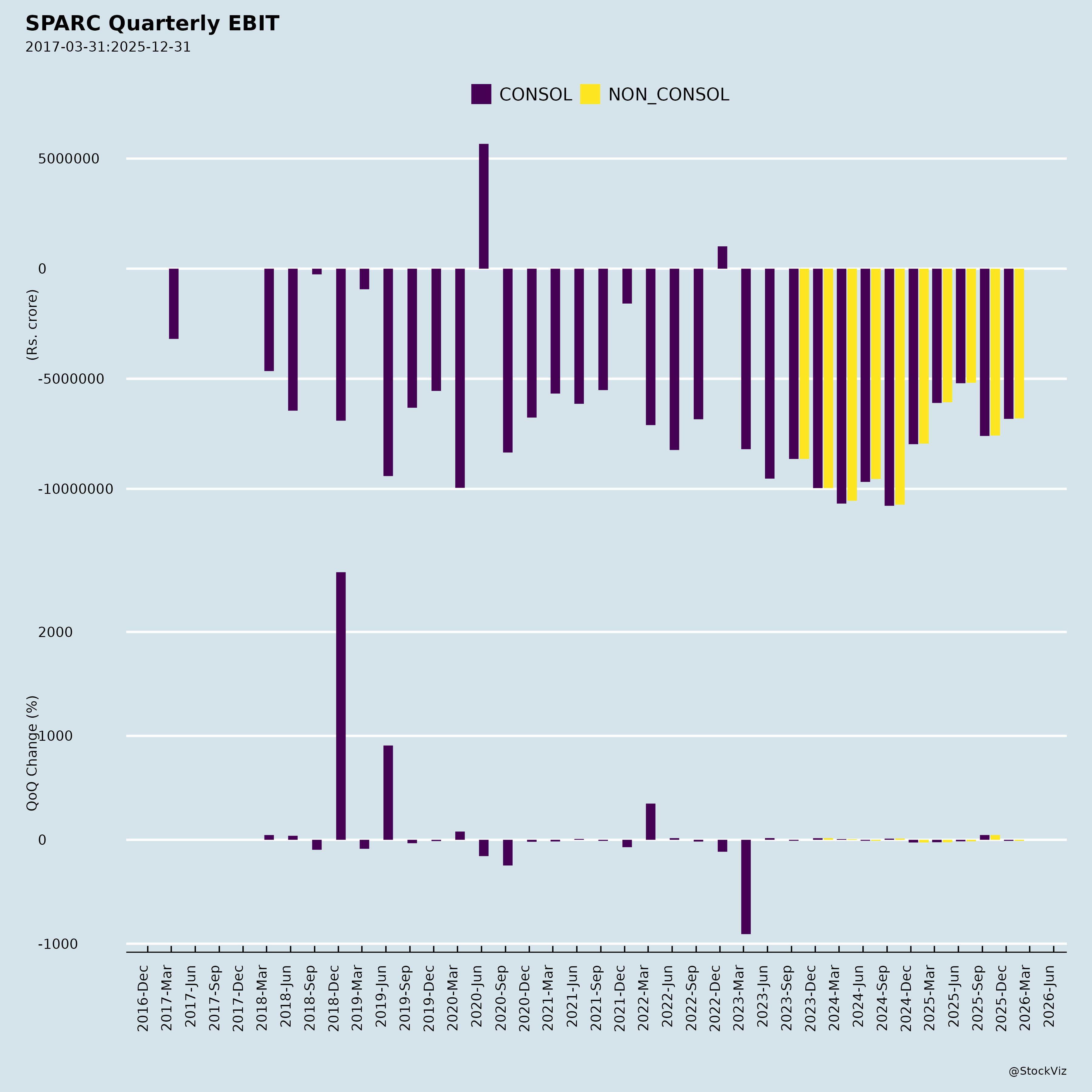
Fundamentals
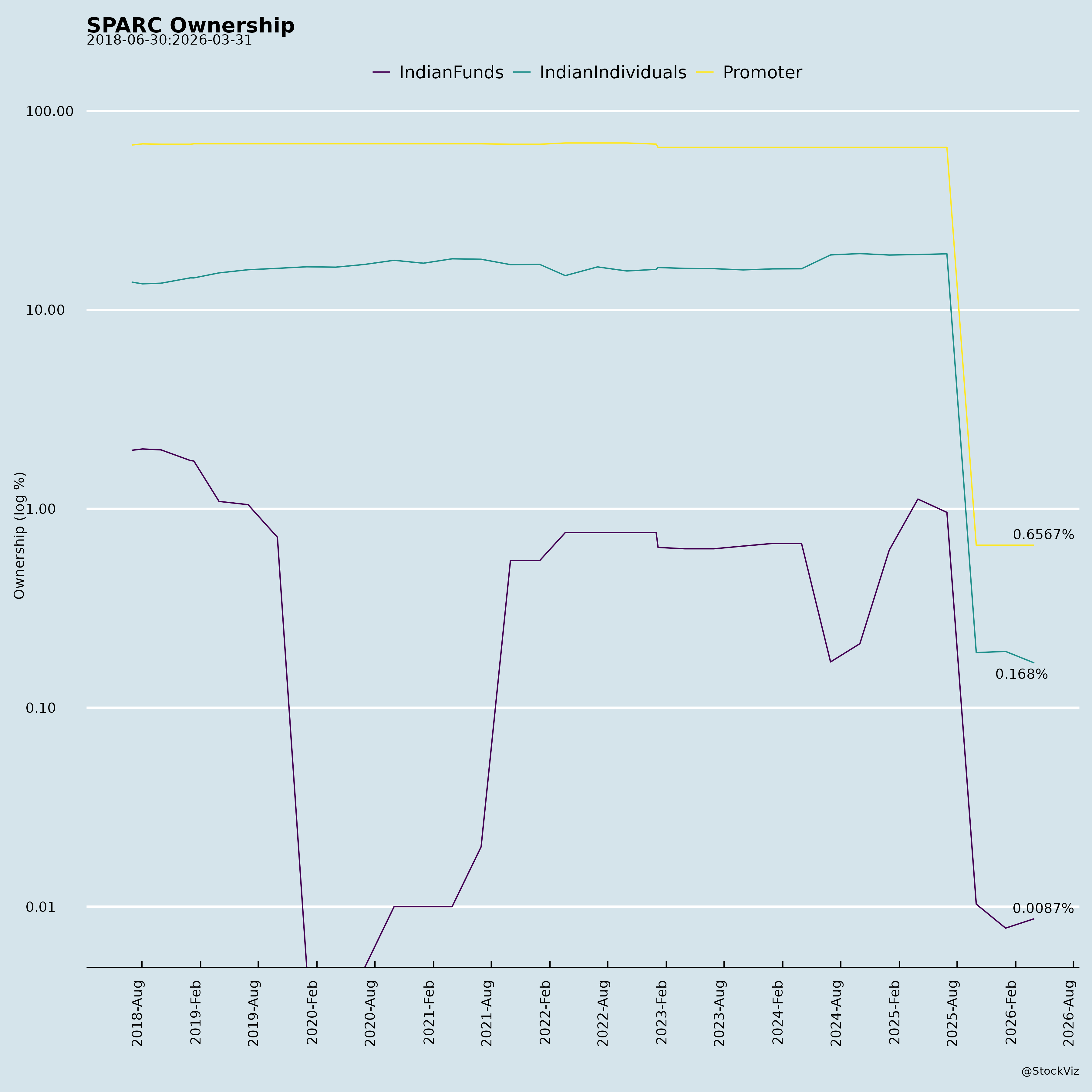
Ownership
Margined
AI Summary
asof: 2025-11-27
SPARC (Sun Pharma Advanced Research Company Ltd.) Analysis
Overview: SPARC is a clinical-stage biotech focused on pharmaceutical R&D (single reportable segment), developing novel therapeutics and delivery tech (e.g., TearAct™). Q2/H1 FY26 (ended Sep 30, 2025) results show ongoing losses amid heavy R&D spend, funded by promoter-linked borrowings. Key pipeline update: Ocuvex resubmitted FDA CRL for PDP-716 (brimonidine ophthalmic suspension), addressing prior facility issues (no efficacy/safety flags). Negative equity and cash burn persist, with going concern tied to promoter support.
Headwinds (Challenges Pressuring Performance)
- Persistent Losses & Declining Revenue: H1 FY26 standalone PBT loss of ₹12,800 lakhs (vs. ₹20,453 lakhs H1 FY25); revenue down 41% YoY to ₹1,750 lakhs. Q2 revenue ₹786 lakhs (vs. ₹1,286 lakhs YoY). Full FY25 loss: ₹34,522 lakhs.
- High Cash Burn: Operating cash outflow ₹11,596 lakhs (standalone H1), driven by R&D/professional fees (₹5,033 lakhs combined) and employee costs (₹5,387 lakhs).
- Debt Burden: Borrowings surged to ₹41,048 lakhs (from ₹25,819 lakhs at FY25-end); finance costs up 741% YoY to ₹1,370 lakhs. Cash equivalents critically low at ₹18 lakhs.
- Negative Equity: Standalone equity -₹34,875 lakhs (vs. -₹22,062 lakhs FY25-end); consolidated -₹34,464 lakhs.
- No Near-Term Profitability: Incurring cash losses quarterly; relies on promoter group support letter for going concern status.
Tailwinds (Supportive Factors)
- Pipeline Momentum: PDP-716 CRL resubmission (Nov 20, 2025) resolves FDA’s July 2023 facility issue—efficacy/safety intact. Potential approval could unlock US market for once-daily glaucoma treatment.
- Promoter Backing: Sun Pharma group provides funding stability (e.g., ₹62,112 lakhs net borrowings inflow H1); intangible assets under development up to ₹7,121 lakhs (standalone).
- Tech Edge: Proprietary TearAct™ platform differentiates products like PDP-716.
- Tax Refunds: ₹3,124 lakhs received H1, aiding liquidity.
Growth Prospects
- High-Upside Pipeline: PDP-716 approval could drive revenue (US ophthalmic market large); other R&D assets in development. New subsidiary Genokine Biotech (Jul 2025) signals expansion.
- Revenue Potential: FY25 revenue ₹7,177 lakhs shows service-based income base; product approvals could shift to royalties/partnerships.
- Strategic Positioning: Sun Pharma ecosystem offers synergies (e.g., commercialization via Ocuvex); focus on underserved ophthalmics/neuro areas.
- Long-Term: If R&D yields 1-2 approvals, could reverse losses (EPS H1: -₹3.94); market cap upside from clinical milestones.
Key Risks
| Risk Category | Details | Potential Impact |
|---|---|---|
| Regulatory | FDA resubmission review outcome uncertain; prior CRL history. Historical trial delays (e.g., high clinical spend ₹1,355 lakhs H1). | Delayed/missed approvals; stalled pipeline. |
| Financial/Liquidity | Cash burn >₹11k lakhs/quarter; debt servicing amid rising rates. Promoter support not guaranteed long-term. | Insolvency risk; dilution via equity raise. |
| Operational | High fixed costs (employees ₹6k+ lakhs H1); execution risks in R&D (intangibles WIP ₹7k+ lakhs). | Further losses; capex strain (₹2,314 lakhs H1). |
| Market/Competition | Biotech volatility; crowded ophthalmics space. Declining service revenue. | Revenue erosion; low commercialization success. |
| Going Concern | Auditor flags dependency on promoter letter; negative equity signals distress. | Trading suspension; value erosion. |
Summary Verdict: SPARC faces acute headwinds from cash burn, debt, and losses, making it high-risk speculative play. Tailwinds from PDP-716 resubmission and promoter support offer catalysts, but growth hinges on regulatory wins (PDP-716 PDUFA timeline TBD). Suitable for risk-tolerant investors eyeing biotech turnarounds; monitor Q3 results/FDA updates. Current setup: High Risk, Binary Outcome Potential. (All figures ₹ in lakhs; consolidated similar to standalone.)
Copyright © 2023 SAS Data Analytics Pvt. Ltd. All rights reserved.